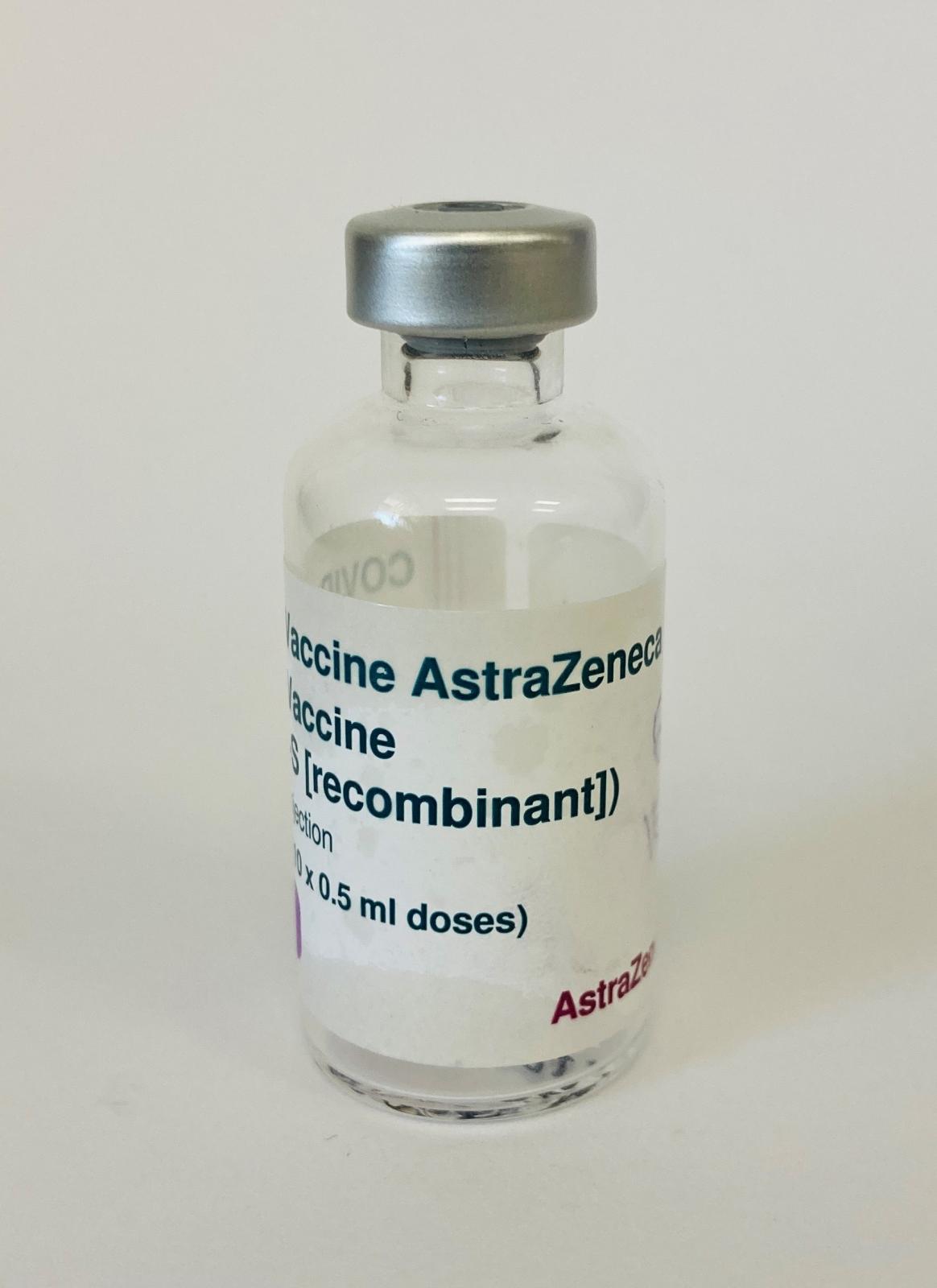
COVID-19 ASTRAZENECA VACCINE PHIAL
2021An empty 5ml glass phial with silver cap and white and purple label with black print reading:
'Covid-19 AstraZeneca, COVID-19 Vaccine, (ChAdOx1-S [recombinant]) solution for i.m. injection, Multidose vial (10 x 0.5ml doses)'.
The purple tag reads '5 ml' in white print.
The lot number '306730P' and expiry date '25 Oct 2021' is printed on the side.
Black handwriting reads '1500' which is the expiry time for drug use. Once the pharmacist reconstitutes the drug (i.e. dilutes with normal saline and vaccine) there is a 6 hour period in which to use the vaccine before it expires.
It is expected to get 10 syringes of 0.5ml of vaccine for administration from one phial.
Australia purchased the Pfizer and AstraZeneca vaccines along with the rights to manufacture AstraZeneca in Australia. A rare side effect of blood clot formation from AstraZeneca vaccines, led to the Pfizer vaccine being initially recommended/administered to all people under 60 years of age.
Details
Details
AstraZeneca
This item is of historical, scientific and social significance. During the Covid-19 pandemic of 2020/21, vaccinations were manufactured world-wide to provide protection to the public from the highly infectious and deadly Covid-19 virus, and to minimise the severity of illness and reduce the need for ventilators and ICU admission.
Other items from Royal Perth Hospital Museum
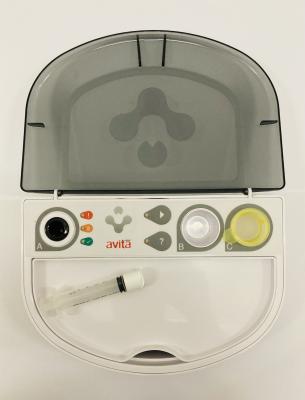
- SURGICAL CAP
- RECELL AUTOLOGOUS CELL HARVESTING DEVICE
- Save RPH Quilt
- Uniform, Nurse
- Operating Table
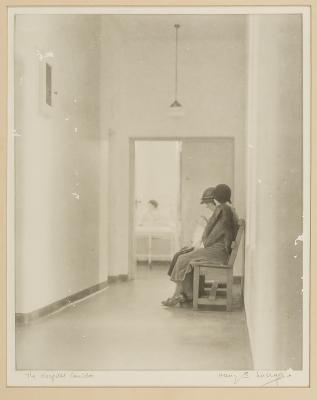
- THE HOSPITAL CORRIDOR, HARRY STEPHENSON LUCRAFT COLLECTION
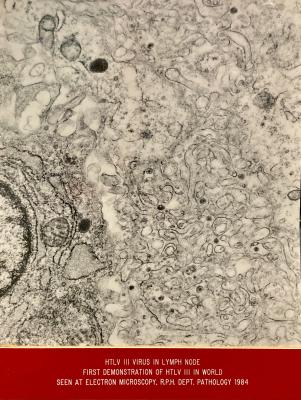
- HIV VIRUS IN LYMPH NODE VIEWED THROUGH ELECTRON TRANSMISSION MICROSCOPE
- Smallpox Camp Subiaco 1893
- PORTRAIT OF DR WILLIAM ELGEE IN FOLDING LEATHER FRAME